Written with Amy Rosenfeld, Ph.D.
By infecting organotypic brain slice cultures from embryonic mice, we have shown that Zika virus has always been neurotropic. The same culture system provides information on how Zika virus infection of the developing brain might lead to microcephaly.
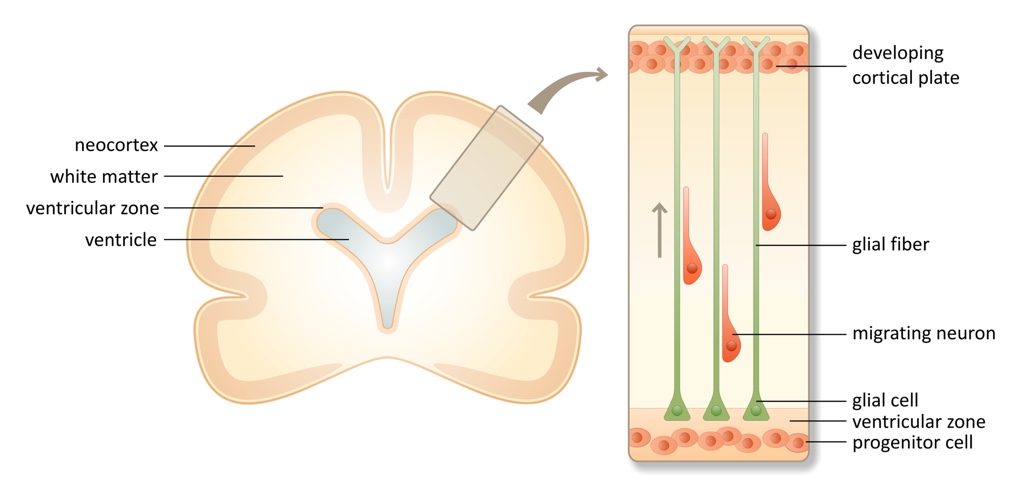
The small heads observed in microcephalic children reflect a physically smaller brain – specifically, the neocortex is thinner than in a normal brain. The neocortex, only found in mammals, is the largest part of the cerebral cortex of the brain. It is composed of six distinct layers of neurons, which are established during embryonic development (illustrated below). First, glial cells originating from progenitor cells in the ventricular zone extend their processes throughout the cortex and anchor at the pia, the outer surface of the brain. These long fibers provide a scaffold on which neurons, produced from the same progenitor cells, migrate outwards to establish the six layers of the cortex. Movies have been made that show the migration of neurons on glial fibers, and they are amazing.
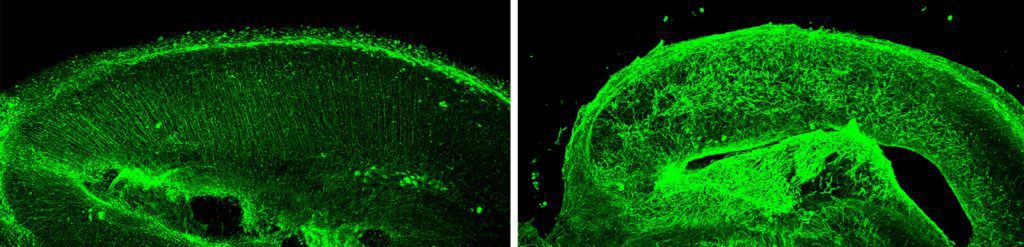
The glial fibers are visible as parallel tracks in our embryonic brain slice cultures stained with an antibody to vimentin, a protein component of the fibers (image below, left panel). When embryonic brain slice cultures were infected with Zika virus, the structure of the glial tracks was altered. Instead of parallel tracks, the fibers assumed a twisted morphology that would not allow neurons to travel from the ventricular zone to the developing neocortex (image below, right panel). Disruption of glial fibers was observed after infection with Zika viruses isolated from 1947 to 2016.

To determine if Zika virus-mediated disruption of glial fibers could impair neuronal migration, we isolated brains from embryonic mice as described above, but before virus infection, a plasmid encoding green fluorescent protein was injected into the ventricle. An electrical current was then applied to the brain to encourage uptake of the plasmid in neuronal progenitors lining the ventricle. The brains were then sliced, placed in culture, and infected with Zika virus.
Four days later, in the uninfected brain slices, green fluorescent neurons could be seen in the ventricular zone, and some of these had already migrated through the developing cortical plate to the pial surface. In brain slices infected with Zika virus, the migration of green neurons to the cortical plate was impaired, and the cells remained in the area where the plasmid was injected. This observation indicates that the disruption of glial fibers caused by Zika virus infection has caused fewer neurons to reach the cortical plate.
We think it is likely that Zika virus disruption of glial fibers during embryonic development contributes to microcephaly: if neurons cannot migrate to the pial surface, the neocortex will be thinner. Zika virus infection also inhibits the proliferation of progenitor cells that line the ventricular surface, which is likely a contributing factor to microcephaly. Other embryonic brain cells are infected with Zika virus, and these could play a role in microcephaly. Furthermore, there are other effects of Zika virus infection on the developing brain, including calcifications, hypoplasia (reduced cell density), lissencephaly (smooth brain), ventriculomegaly (enlarged ventricle), and brainstem dysfunction.
We are particularly interested in identifying the viral protein that disrupts the glial fibers in embryonic mouse brains. Once that protein is identified, it might be possible to understand the mechanisms by which the glial fibers are disrupted. Such information would not only lead to a better understanding of how Zika virus causes microcephaly, but should also provide a better understanding of how the brain develops.

Thanks for the interesting post! That protein may be NS2A. See the article “Zika-Virus-Encoded NS2A Disrupts Mammalian Cortical Neurogenesis by Degrading Adherens Junction Proteins”.
I’ve always wondered how it is that glial cells can find their way on their own, but neurons can’t. Evolution really should have combined them by now, and then we could fit much more data into the space! 🙂
Pingback: TWiV 468: Zika by the slice | This Week in Virology
Pingback: TWiV 468: Zika by the slice – Atomic Bombs and Mermaids