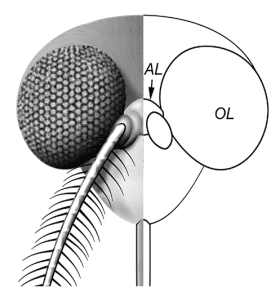

Mosquitoes are vectors for the transmission of many human viral diseases, including yellow fever, West Nile disease, Japanese encephalitis, and dengue hemorrhagic fever. Many mosquito-borne viruses enter the human central nervous system and cause neurological disease. In contrast, these viruses replicate in many tissues of the mosquito, including the central nervous system, with little pathological effect and no alteration of behavior or lifespan. The defenses that allow such persistent infection of mosquitoes are slowly being unraveled.
A protein called Hikaru genki, or Hig, is crucial for controlling viral infections of the mosquito brain. Originally discovered in the fruit fly Drosophila, Hig is produced mainly in the brain of Aedes aegyptii, the natural vector for dengue and yellow fever viruses. Experimental reduction of Hig mRNA or protein in the mosquito leads to increased replication of dengue virus and Japanese encephalitis virus. This increase in viral replication is accompanied by more cell death in the mosquito brain, and decreased survival.
How does Hig protein impair virus replication? The Hig protein of A. aegyptii binds dengue virus particles via the E membrane glycoprotein. As Hig protein is located on the cell surface, binding to virus particles prevents virus entry into cells. Impairment of endocytosis is limited to insect cells – introduction of Hig into mammalian cells had no effect on virus replication. Clearly other components of insect cells must participate in the Hig-mediated antiviral mechanism.
The antiviral activity of Hig protein depends on the presence of sushi repeat domains, also known as complement control protein (CCP) domains. These consist of 60 amino acid repeats with four conserved cysteines and a tryptophan. The CCP domain is found in many proteins of the complement system, a collection of blood and cell surface proteins that is a major primary defense and a clearance component of innate and adaptive immune responses. The sushi domain mediates protein-protein interactions among complement components. Capturing the dengue and Japanese encephalitis viruses by the A. aegyptii Hig protein is just one example of the virus-binding ability of proteins with CCP domains. An insect scavenger receptor with two CCP domains is a pattern recognition receptor that recognizes dengue virus and recruits mosquito complement to limit viral replication. Some CCP containing proteins are virus receptors (complement receptor 2 binds Epstein-Barr virus, and membrane cofactor protein is a receptor tor measles virus).
Because the Hig antiviral machinery is largely limited to the mosquito brain, it is possible that it prolongs mosquito life to allow virus transmission to other hosts. Transmission of virus to other hosts requires replication in the salivary gland, which cannot take if the mosquito dies of neural infection. I wonder why humans do not have have similar mechanisms to protect their neural tissues from virus infections. Is neuroinvasion a less frequent event in humans, compared with mosquitoes, thereby providing less selective pressure for protective mechanisms to evolve?

Well done. Has anyone looked for similar mechanisms in the human brain, or in mammalian brains?
Hig protein is not found in vertebrates. It would be interesting to look for any sushi containing proteins in the human brain. I’m not aware of any such studies. Unlike mosquitoes, humans are harmed by viruses replicating in the brain.
I am assuming that this has nothing to do with the sushi you get at a restaurant. Sushi is just related to the name it is called. Kind of like Sonic Hedgehog?
HIV in mosquitoes? There was a time when mosquitoes were suspected of
transmitting HIV. None less than the 2008 Nobel Laureate Françoise
Barré-Sinoussi and her colleagues reported in 1986 that “HIV proviral
DNA has been also found in various insects from Central Africa (Zaire
and Central Africa Republic) but not similar insects from the Paris
area. These data suggest that insects could be a reservoir or a
vector
for the AIDS virus” [1]. As a student of bioinformatics I was curious
to see if there were more recent data to substantiate this report. Using
TBLASTN the HIV-1 protein sequences published at the Los Alamos HIV
Database were tested against translated mosquito DNA sequences. https://goo.gl/7NydgO The alignments were interpreted according to the Doolittle rules of
thumb [2, 3]. Sequences with E-values =25% are
considered significant and may point to protein similarity. They may
also represent potential immunogens. As a “control†I searched for
similarities between mosquito proteins and proteins of RNA viruses with a genome similar in length to HIV-1. However, none were found. Although HIV, AIDS, mosquitoes and Africa are linked, it is nonetheless difficult to interpret these findings. They could be a mere coincidence but if not it is a mystery why the genome of mosquitos should contain HIV-1 gag and pol sequences. Interestingly, in searches involving other taxa I
have also discovered alignments with the HIV-1 proteins [4]. Perhaps some readers could confirm my findings. I welcome opinions others may have to explain these data.
1.Becker JL, Hazan U, Nugeyre MT, et al. Infection of insect cell lines
by the HIV virus, an agent of AIDS, and a demonstration of insects of
African origin infected by this virus. C R Acad Sci III 1986; 303:303-6.
http://www.ncbi.nlm.nih.gov/pu…
2. Kyte J, Doolittle RF. A simple method for displaying the hydropathic character of a protein. J Mol Biol 1982; 157:105-32.
3. Lesk A. Introduction to protein science: architecture, function, and genomics: Oxford University Press; 2010.
4. Findings: https://goo.gl/dc5R0R