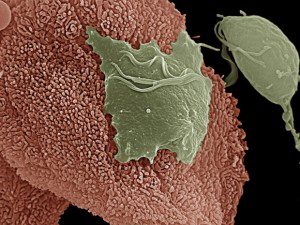

Trichomonas vaginalis is a protozoan parasite that infects the genitourinary tract of ~250 million individuals each year, leading to bacterial vaginosis, increased susceptibility to human immunodeficiency virus (HIV) and human papillomavirus (HPV), and reproductive complications such as infertility, pregnancy loss, and preterm delivery. Antibiotic treatment can clear the parasite but does not prevent some of these complications.
Half of T. vaginalis clinical isolates harbor dsRNA viruses (TVV) that do not harm their parasite host. It has been suggested that these TVVs are sensed by the human host, leading to inflammation and reproductive complications. To test this hypothesis, human epithelial cell cultures from the female reproductive tract were exposed to TVV-negative or positive T. vaginalis. Virus-positive parasites induced the production of interferon and proinflammatory cytokines while virus-negative parasites did not. The production of these cytokines is dependent upon Toll-like receptor 3 (TLR3), which is present on the inner endosome membrane. This observation suggests that virions or viral RNA released from T. vaginalis are taken into the cell by endocytosis where they encounter TLR3. In support of this idea, the addition of purified TVV virions to cells lead to TLR3-dependent production of cytokines.
To provide a plausible explanation for the failure of antibiotics to prevent T. vaginalis associated complications, protozoans with or without TVV were treated with metronidazole for 24 h, and the cell supernatant was added to cell cultures. Supernatants from the virus-containing, drug treated protozoa induced cytokines in a TLR3-dependent manner. Cytokines were not induced using supernatants of T. vaginalis lacking TVV, or from cells that were not treated with antibiotics. These results suggest that antibiotic treatment of virus-infected T. vaginalis causes an inflammatory response by liberating TVV virions or viral dsRNA from the protozoan.
These intriguing results show that protozoan viruses can be sensed by human cells, leading to an inflammatory response. In the vaginal mucosa this response is a double-edged sword: it can help limit infection but may also lead to pathological damage and increased susceptibility to bacterial and viral infection. Clinical studies are needed to determine if inflammation caused by TVV is linked to T. vaginalis associated disease and complications. The outcome of this work may determine whether the use of antivirals to inhibit TVV replication, or anti-inflammatory agents, should accompany antibiotic therapy to prevent the complications associated with T. vaginalis infection.

I may have missed a step, but how is it that a protozoan “leads to” *bacterial* vaginosis?
There doesn’t say so, just says that inflammation can cause the body to be more susceptible to get bacterial or viral infection.
Would the same kind of reaction also apply to bacterial prophages, or no?
Pingback: TWiV 212: Apocalypse TWiV 122112 212
Pingback: A virus in a parasite in a human