
Rotaviruses are the single leading cause of diarrhea in infants and young children. Each year rotavirus gastroenteritis causes over 1,250,000 episodes of diarrhea and 527,000 deaths, mainly in developing countries. Rotavirus vaccines are used to reduce the global burden of rotavirus disease. Rotarix, the vaccine manufactured by GlaxoSmithKline, is an infectious, attenuated vaccine that is administered orally to infants.
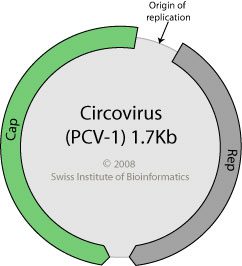
The genetic information of rotaviruses consists of 11 segments of double-stranded RNA. In contrast, porcine circoviruses are small viruses with a circular, single stranded DNA genome (pictured). At 1.7 kb in length, the DNA is among the smallest known viral genomes and encodes only two proteins. Porcine circovirus 1 was originally discovered as a contaminant of a pig kidney cell line. Later a second strain, porcine circovirus 2, was isolated and shown to be associated with postweaning multisystemic wasting disease, an emerging disease of swine.
Porcine circovirus 1 DNA was also found in the cells used to produce Rotarix. Therefore the contaminant has been present since the early days of vaccine development, including clinical trials. A different rotavirus vaccine produced by Merck, called RotaTeq, does not contain porcine circovirus DNA.
It’s not at all clear that the presence of porcine circovirus 1 DNA in Rotarix is a problem. Circoviruses have not been associated with human disease, and porcine circovirus 1 has not been found to cause disease in any animal. Many humans have antibodies to these viruses, including porcine circovirus, indicating that they were infected at one time. Furthermore, it’s not known if the vaccine contains infectious virus or DNA fragments. Nevertheless, it’s always preferable to err on the side of caution, as the FDA has done.
Because porcine circoviruses are widespread in commercial swine populations, there have been concerns about the use of porcine organs for xenotransplantation and for the production of products used in humans such as factor VIII, heparin, insulin and pepsin. It is therefore important to ensure that such products do not contain infectious circoviruses.
This incident will undoubtedly further increase public distrust of vaccines and vaccine manufacturers. I have not yet seen the journal article describing these findings, so I can’t comment on why the contaminant was not identified early in vaccine development. According to the FDA, “In four to six weeks, FDA will convene an expert advisory committee and make additional recommendations on the use of rotavirus vaccines.” If Rotarix is found to contain infectious porcine circovirus, then its use will certainly be discontinued. However, detection of small noninfectious fragments of porcine circovirus DNA Rotarix will likely lead to resumption of vaccine use. In either case, new lots of vaccine should be produced using circovirus-free cells.
Tischer I, Bode L, Apodaca J, Timm H, Peters D, Rasch R, Pociuli S, & Gerike E (1995). Presence of antibodies reacting with porcine circovirus in sera of humans, mice, and cattle. Archives of virology, 140 (8), 1427-39 PMID: 7544971
Fenaux M, Opriessnig T, Halbur PG, Xu Y, Potts B, & Meng XJ (2004). Detection and in vitro and in vivo characterization of porcine circovirus DNA from a porcine-derived commercial pepsin product. The Journal of general virology, 85 (Pt 11), 3377-82 PMID: 15483254

“an independent research group found that the vaccine contains DNA of porcine circovirus type 1”
Why was this not checked before? Such thing as vaccines should be tested to a perfection level.
I fully agree. However, in the case of vaccines, perfection is limited
by the experimental techniques available. Apparently a novel technique
was used to detect circovirus DNA which previously had not been found.
We'll have to wait for publication of the manuscript to learn more
about exactly what was done.
<< This incident will undoubtedly further increase public distrust of vaccines and vaccine manufacturers. >>
Rightfully so. But I think you're missing the bigger picture.
The big picture for a growing number of concerned parents, including myself, is that public health experts and doctors are pretending to have certainty in the safety of vaccines that science simply does not have. This is just another example of uncertainty and unknowns.
We want honesty about the risks and the unknowns. We don't want to be pressured to give our children the full schedule. Instead, we weigh the risks/benefits of each vaccine for our children. What we hear from pediatricians does not match what we see in the real world. Rotavirus is not a big deal in developed nations. I’d rather that my kids get real immunity from the real flu than annual flu shots. H1N1 was a joke and left many more parents doubting public health experts than ever before. And then there is HepB. That is what started me down the path of questioning the recommended schedule. There was zero chance that my newborns were at risk and yet they were supposed to get the HepB shot? Really smart doctors were giving me reasons for it that sounded dumb and irrelevant.
Pressure and fake certainty (PR/lies) creates resistance and distrust. Honesty and persuasion is the more intelligent route.
Thank you for an interesting and informative article.
Here is a recent interview that provides a powerful perspective about wanting honesty about risks: http://tinyurl.com/yhprv7f
“This incident will undoubtedly further increase public distrust of vaccines and vaccine manufacturers. I have not yet seen the journal article describing these findings, so I can’t comment on why the contaminant was not identified early in vaccine development. “
XMRV was discovered in 2006. How do we know if a vaccine is contaminated if we don't even know what to look for?
http://www.retrovirology.com/content/7/1/16 reports about the possible dangers of XMRV transmission in transplant patients for example.
Because we don't know about certain viruses, could it be that we actually transmit them first through vaccines, blood transfusions etc.?
Just out of curiousity, how would one go about determining whether a lot of vaccine contains an infectious virus? One could go the qPCR route, but that's a huge world of viruses to test and it doesn't preclude undiscovered viruses or mutant viral genomes that don't bind your primers. ELISA would have a similar problem. I also thought of plaque assays but they probably depend on cytopathic viruses. Anyway, I'm not too surprised this happened, given it's happened historically with SV40 (and bacteriophage, but they don't count) and the polio vaccine. Completely defining products made from cell culture, it seems to me, is always a hard business.
I believe that the best way is to use a pan-viral microarray. Extract
nucleic acid from a vaccine preparation and hybridize it with an array
containing sequences from every known virus. The limitation is that
you can only test for the viruses you know about. For this reason,
there will likely always be incidents like this until we no longer use
animal cells to produce vaccines. Perhaps we should be using vaccines
produced in plants?
It might be hard to express antigens correctly in plants given that they're far way phylogenetically. For instance, they're not exothermic organisms and that might affect protein folding. That said, if it's just subunit vaccines we're talking about, I'm sure irradiating the poop out of the vaccine lot should solve any virus lurkers.
But what do I know, I'm not in the business of making vaccines!
How about mass sequencing? You don't need to know what you're looking for, and the price is perhaps reasonable enough now to consider doing it for each production lot.
Sensitivity may not be high enough though. If you have a 0.001% contaminant it might not be picked up. On the other hand perhaps it could be used to check whether the cell line used for production secretes some unknown virus before you start production.
DMc
This virus was known so would have been caught with such an approach. Clearly we can never have the certainty that the parent above wants. (I, too, am a parent – I just also happen to be a scientist so I understand that there will always be uncertain in the world.) As soon as we produce vaccines in plants, there will be worries about plant interactions, allergies, etc.
Actually your idea isn't a bad one! I think the sensitivity will be fine because there are amplification steps to high throughput sequencing. If anything, I'd think it'd be too sensitive.
If we weren't all in this together I'd be happy to let you risk your kids health. But we are (Google “herd immunity”. Life is full of uncertainty and unknowns. Science can never, ever tell us everything with perfect certainty. But it can tell us more than Jenny McCarthy. Nobody is pretending to have certainty except for folks like Mrs. McCarthy: many huge, comprehensive and expensive experiments have been carefully done that fairly clearly defines the risk. It is clear that many fewer people are dying from many viral diseases, and in the future from liver, cervical, vaginal and anal cancers.
You're getting honesty about the risks and unknowns. No one knew this viral nucleic acid was in this preparation. Now it's been found and reported. There is no evidence that it causes any harm and lots of work will follow up to find out. This is in contrast to the diseases that have been prevented by vaccines. Google images for smallpox. Are you old enough to remember iron lungs? These are known and certain.
We are lucky that H1N1 didn't kill more people. We have to be vigilant when something like this happens – no one knows for sure what will happen. Attitudes like yours don't help. You're likely the type that'd be yelling loudest if it ended up a massive killer. Where were the experts?!? There is no pressure and fake certainty (lies?) – please remove the tin foil and take a deep breath.
People get sick and die (of unknown causes) without the Gardasil vaccine too. This is precisely why studies are done to exclude these possibilities: you compare a vaccinated group to a control group.
I feel sorry for people that have lost a child and want an explanation. But rejecting vaccines for the wrong reasons will lead to orders of magnitude more loss of life. Coincidences occur, and grieving parents need something to blame.
You were doing a good job, Sciguy, until your last paragraph when you resorted to the overused 'tinfoil hat' slur. I really wish more scientists had the class that Dr. Racaniello has. I don't agree with every position Dr. R takes, but I respect him and return to read his blog and am not afraid to recommend it to others.
The FDA did the right thing making the recommendations it did regarding Rotarix. People should at least take comfort from that. Now when will the cell culture mess get straightened out so this, (or something worse), does not happen again?
http://digitalmedia.cua.edu//events/video/asx_d…
http://cellbank.nibio.go.jp/cellbank/qualitycon…
So you didn't mind the Jenny McCarthy part, Scigal? Well, let me first say that I wish I had the class of Dr. Racaniello too: my passion can be a personal failing. It is hard not to become emotional when you've worked hard, long hours in the lab for most of your life, not making a tremendous amount of money. Most scientists do this because we LOVE what we do. I consider it a personal assault to read people accusing science of systematic lies. I understand that many people have trouble with coincidence and cause and effect, but I honestly consider these types of conspiracy accusations baseless lunacy. I am tired of this aggressive activism and I think that we (scientists) need to debate these arguments openly instead of just being quiet and avoiding conflict. That's how we've ended up where we are in the first place. This blog and podcast have already created an environment where we (scientists and laypersons) can interact and I think it's great. But I think it's a mistake to shrink away from our point of view because someone is louder and bullying.
I agree with you that the FDA did the right thing, and I think we should all work as hard as we can to make sure that it doesn't happen again. But it's important to recognize that it will, and as pointed out there can never be 100% certainty. We (scientists and regulators) should strive for perfection but you (laypeople) should understand that perfection can never happen. Accidents will happen and there will always be viruses yet to discover. The sooner we can all come to this realization, the sooner we can work together to keeping all of our kids safe. It doesn't help us (scientists) to have to convince people that vaccines are a good thing. I'll do my best to keep my emotions in check and the debate in the ring.
Sciguy, I haven't studied Ms. McCarthy's situation and beliefs in depth so don't have an opinion there and don't think you were rude towards her. I just didn't see Chris as attacking scientists in general. He describes a dispute he had with his personal doctor over one vaccine. Maybe he was a little negative overall because of that.
I can understand feeling reactive, though, and I appreciate your honesty about nothing being perfect. I believe in vaccines being voluntary, but I do see their value. The human immune system is not perfect, either. Even if you have a good one; stress or chemical exposures or even grief can take it down. If you get sick on top of that, it can be devastating. We aren't in control of everything that happens to us. So I wouldn't worry that people will give up on vaccines as prevention.
The novel H1N1 outcome even made me wonder about creating replacement viruses. I wonder if it would ever be possible to make a flu virus that could ecologically replace virulent viruses, without it harming people.
Sciguy, I haven't studied Ms. McCarthy's situation and beliefs in depth so don't have an opinion there and don't think you were rude towards her. I just didn't see Chris as attacking scientists in general. He describes a dispute he had with his personal doctor over one vaccine. Maybe he was a little negative overall because of that.
I can understand feeling reactive, though, and I appreciate your honesty about nothing being perfect. I believe in vaccines being voluntary, but I do see their value. The human immune system is not perfect, either. Even if you have a good one; stress or chemical exposures or even grief can take it down. If you get sick on top of that, it can be devastating. We aren't in control of everything that happens to us. So I wouldn't worry that people will give up on vaccines as prevention.
The novel H1N1 outcome even made me wonder about creating replacement viruses. I wonder if it would ever be possible to make a flu virus that could ecologically replace virulent viruses, without it harming people.
Pingback: Rotavirus vaccine and herd immunity | Mystery Rays from Outer Space
Pingback: A plant virus that switched to vertebrates
Pingback: Porcine circovirus DNA found in RotaTeq
I did not most children are vaccinated today,and I am still healthy.I assume that the health sector tells you to jump, you jump.
xanax online overnight shipping
Can’t wait to see the manuscript!Really interesed
Pingback: ×בולוציה בפעולה: × ×’×™×¤×™×, ×¦×ž×—×™× ×•×’×•×¨×™ ×›×œ×‘×™× | מגירה 2.0
Pressure and fake certainty (PR/lies) creates resistance and distrust. Honesty and persuasion is the more intelligent route.Â
The blog was absolutely fantastic! Lots of great information and inspiration, both of which we all need!b Keep ’em coming… you all do such a great job at such Concepts… can’t tell you how much I, for one appreciate all you do! cash advances
 Hi, your blog has helped me a lot in researching on this topic. It sure makes a lot of sense. Please keep updating this page, since I intend to visit it much more often now. Thank you!