By Gertrud U. Rey
Gertrud Rey is a trained virologist residing in Atlanta, Georgia. During the day, she works as a consultant in a biotech patent law firm, but spends much of her free time as a science communicator. She was a guest on TWiV 179 and 424.
Although the dominant mode of transmission for influenza virus is via the respiratory route, there is growing evidence to suggest that various human and avian influenza viruses are capable of entering the body following ocular exposure.
Influenza infection of the eye may be established locally, manifesting itself in the form of conjunctivitis, or it may spread to the respiratory tract via the nasolacrimal duct, which links the eye to the nasal passages. Influenza viruses which are more predisposed to infect via the ocular route and cause ocular symptoms, are termed “ocular tropic.” Most cases of ocular influenza virus infection occur during the process of culling infected poultry. As this procedure often involves handling of contaminated material and the formation of aerosols, workers who don’t wear proper protective eyewear, or who touch their eyes with contaminated hands, are most likely to be infected.
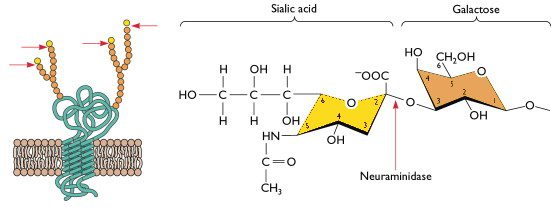
Influenza virus host range and tropism are thought to be controlled by the cellular receptors present in the human respiratory tract. Human influenza viruses preferentially bind sialic acid receptors (illustrated), which are linked to galactose via an alpha 2,6 glycosidic bond, while avian viruses preferentially bind sialic acids with an alpha 2,3 linkage (for a review of how influenza virus attaches to cells, click here).

A recent study by Hannah Creager and colleagues at the Centers for Disease Control and Prevention analyzed potential determinants of ocular tropism, including:
- different methods of inoculation (aerosol versus liquid);
- cultures that were grown at different temperatures (body temperature versus 33°C)
- cultures that were of varying complexities (cell monolayers versus corneal tissue explant cultures)
- factors external to cells, such as the interaction between influenza viruses and human tears
First, Creager analyzed the ability of both ocular tropic and non-ocular tropic viruses to replicate in primary human corneal epithelial cells. She found that viruses isolated from the respiratory tract of human influenza cases replicated efficiently in these cells, to a level comparable to that observed with ocular tropic viruses isolated from swabs of human conjunctivitis cases. This observation suggests that corneal epithelial cells are permissive to both ocular and non-ocular influenza viruses.
The next set of experiments took two factors into consideration: infection via aerosol, and temperature. In contrast to the core body temperature of 37°C, both the ocular surface and the upper respiratory tract temperatures are about 33°C. To determine whether ocular viruses are better at infecting via aerosol, and whether they might replicate more efficiently at the lower temperatures of the human ocular surface, corneal cell monolayers were exposed to different aerosol doses of both ocular and non-ocular tropic viruses (using liquid infections as a control). After this exposure, half of the cultures were incubated at 37°C, and the other half at 33°C. The results suggest that neither the method of inoculation, nor the temperature at which cells were cultured prevented viruses isolated from the respiratory tract from establishing a productive infection in human corneal epithelial cells.
As a typical monolayer cell culture does not represent an authentic setting for ocular viral infection, liquid infection experiments were repeated in corneal tissue explant cultures. These tissue cultures consist of a stratified epithelium with approximately five layers of cells. They are grown at an air-liquid interface, and since they include tight junctions, various mucins, and a higher presence of alpha 2,3-linked sialic acids compared with alpha 2,6-linked sialic acids, consistent with the epithelial cells of the conjunctiva, they tend to provide a highly predictive non-animal alternative to assess various aspects of infection and disease modeling. Basically, they make a good in vitro laboratory model of the human eye. Infection of these cultures with both ocular and non-ocular tropic viruses showed that ocular tropic viruses infect eye cells more efficiently than do non-ocular tropic viruses.
In addition to providing lubrication, tears are known to protect the eye from infectious agents, and present a notable barrier to viral entry. In a final set of experiments, Creager examined the effect of tears on viral viability using a plaque reduction assay. Briefly, this assay involves mixing equal volumes of either virus and tears, or virus and saline; incubating the mixtures for one hour at room temperature; and then plating them on cells in accordance with a standard plaque assay. Viral infectivity is then calculated by dividing the number of plaques on each tear-treated well by the number of plaques counted on its corresponding saline-treated well. The results show that infection by ocular tropic viruses was less inhibited by tears than infection by non-ocular tropic viruses, and that multiple tear components contribute to this inhibitory effect.
There are several good reasons why continued research in this area of influenza virology is needed. Although conjunctivitis from influenza infection is pretty rare, the ability of some influenza viruses to enter the body via the ocular route does present some concern, particularly if one takes into consideration the potential for spread to the respiratory tract via the nasolacrimal duct. Furthermore, since the eye mucosa is immunologically similar to other mucosal compartments, eyedrops present an attractive alternative vaccine strategy against influenza virus infection that would help overcome some of the challenges presented by conventional vaccine methods.
For a deeper understanding of ocular infection by influenza virus, I recommend this article by Jessica Belser, published in The Lancet Infectious Diseases.

Pingback: Influenza virus in the eye - Vetmedics