
To answer this question, ten genetically marked polioviruses were mixed and subjected to plaque assay. Of 123 plaques, 6 (4.9%) contained more than one virus. Similar results were found when polioviruses with phenotypic markers were studied.
Examination of poliovirus stocks by electron microscopy revealed both single particles and aggregates of 2 to 10 particles. Increasing particle aggregation by treatment of viruses with low pH increased co-infection frequency, indicating that aggregation of particles leads to multiply infected cells.
When these experiments were repeated with mutagenized polioviruses, the co-infection frequency increased – probably because recombination and complementation between two defective genomes leads to rescue of the defects.
Do these findings indicate that poliovirus plaque formation does not follow one-hit kinetics? The results do not prove that, in unmutagenized virus stocks, more than one poliovirus is needed to form a plaque. They only show that a small percentage of plaques contain more than one poliovirus. The presence of more than one poliovirus in 5-7% of plaques is likely a consequence of virion aggregation. It would be informative to prepare poliovirus stocks with no aggregates, and determine if co-infected plaques are still observed.
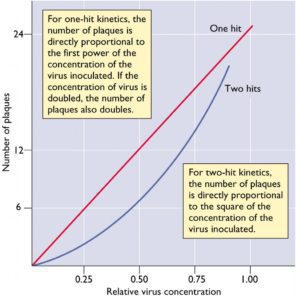
Some viruses of plants and fungi follow two-hit kinetics: two virus particles, with two different genomes, are needed for infection (illustrated). Assuming that 4-7% of poliovirus plaques are initiated by multiple viruses, the resulting plots deviate only slightly from a straight line, and do not resemble the curves of two-hit kinetics.
What are the implications of these findings for the use of plaque assays to produce clonal virus stocks? Even though the frequence of multiply infected plaques is low, the possibility of producing a mixed population is still possible, if only one plaque purification is done. In our laboratory we have always repeated the plaque purification three times, which should ensure that no multiply infected plaques are isolated.
Update 3/31/17: I would like to see similar experiments done with other viruses, to see how often multiple viruses can be found in a plaque. Examples included hepatitis A virus, which is released from cells in membranous vesicles containing multiple virus particles; and enveloped viruses, which might aggregate more frequently than naked viruses.
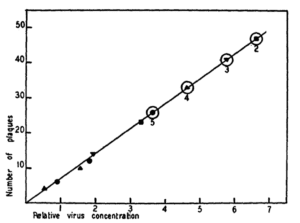
I looked back at the 1953 publication in which Dulbecco and Vogt first described the plaque assay for poliovirus, and demonstrated one hit kinetics. The dose-response curve clearly shows one-hit kinetics with little deviation of the individual data points from a straight line.


Pingback: fungus | [Veterinary and Medical Sciences
Pingback: The purity of plaques – Virology