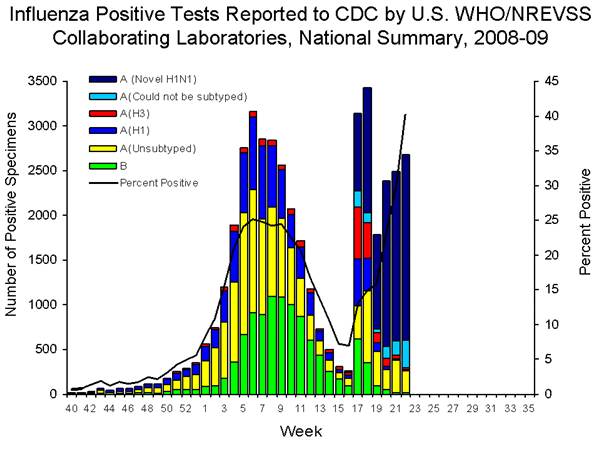
A network of ~150 laboratories in the United States participate in virological surveillance for influenza. The results of these analyses are publicly available, and provide extremely interesting characterization of the pandemic spread of the new H1N1 strain. During week 22, 6,664 samples were submitted for testing, of which 2,681 were positive for influenza virus. Of these, 2,071 (89%) were identified as the new H1N1 strain. The previous seasonal influenza A (H1), A (H3), and B viruses continue to co-circulate. These observations are summarized on this graph:
I have noted previously the unusual upsurge in influenza activity during weeks 17 and 18, which coincided with the emergence of the new H1N1 virus. The number of virus-positive specimens dropped during week 19, but has since risen. More importantly, the percent positive specimens – the black line – continues to rise. This observation suggests that the new H1N1 virus will continue to circulate, at least for the next month. Increased sampling by clinicians in the face of a pandemic probably also contributes to the rising numbers of influenza positive specimens.
Also of interest is the declining circulation of the two influenza virus strains of the previous season – H3N2 and H1N1. Of the 2,681 influenza positive samples in week 22, 9 were identified as the previous seasonal H1N1 strain, and 22 as the H3N2 strain. The circulation of these seasonal influenza viruses into June is probably not abnormal. Because of the pandemic H1N1 strain, more samples are being tested than is usual. Clinicians stop testing for influenza virus at the end of May as the ‘flu season’ wanes. CDC stops its influenza surveillance in May, and resumes in the fall. This year CDC will probably keep up the influenza reporting through the summer. According to Dr. Anthony Fiore, a medical epidemiologist at CDC,
Now people who don’t normally look for flu are looking more than they ever have. We’re seeing that some of the respiratory illnesses that occur even late in the season may be due to seasonal flu viruses that we didn’t appreciate in the past. I guess we don’t know that anything different is occurring right now with seasonal flu viruses. This is an artifact of clinicians looking very hard for flu viruses at a time they don’t normally look.
The textbooks have always said that influenza viruses can be isolated from large northern hemisphere cities throughout the summer. The increased surveillance this year will provide a more detailed picture of the extent of influenza virus spread.
I’m also looking forward to reviewing influenza surveillance data from the southern hemisphere in the coming months. The Department of Health and Ageing of Australia provides similar data as the CDC, but their information has not been updated since 22 May and consequently the impact of circulation of the new H1N1 strain is not evident.

Very interesting. At what point do we just consider the 2009 H1N1 the “seasonal H1N1 strain†(supplanting the previously circulating H1N1 strain)?
The entire dominate stain theory is quite interesting – I guess its all about host vulnerability and the antigenic variation of an ever-changing virus?
The 2009 H1N1 strain becomes a 'seasonal H1N1 strain' when there are
no longer global epidemics, just local epidemics. Probably after two
seasons of pandemic spread. The supplanting of strains is puzzling –
one theory is that the pandemic strain induces 'memory' antibodies in
the host that get rid of the older strains.
that's interesting … make a new blog-entry about it ?!?
We also have H3N2-seasons and H1N1-seasons, but rarely both.
Maybe Mexicanflu,Hongkongflu,Russianflu rotational in future (plus B).
The more strains the less likely a new pandemic, hoping that they won't
add but “fight” each other
I see what you mean about the puzzle of one flu displacing another. Unless there is some sort of interaction- a mechanism whereby infection with a novel flu somehow alters the outcome of a subsequent (potential) infection by an established seasonal flu then there is no competition between them- and one would expect the seasonal flu infection to simply continue in parallel with the novel flu.
Could it be that the action is not in the experienced hosts at all, but in the naive hosts? While I understand that antigenic drift can help keep a seasonal flu going in a population that has already developed a significant level of immunity, it must also surely be the case that the constant supply of totally naive hosts (new humans) must make an important contribution to keeping any seasonal influenza going (the naive hosts must make a contribution to R0, and if a seasonal flu is just bumping along in equilibrium then its R0 must be quite near the edge?). What if there is a blanket reduction in susceptibility to *any* kind of flu infection once an individual has had their first ever flu infection, and/or, perhaps such naive hosts are extra infective during their first ever flu infection (or at least more infective, all other things being equal, than they would be for any subsequent flu infection). If this were the case then, as a novel flu, you would indeed be ‘taking something away’ from the existing seasonal flu if you can be the first to infect a completely flu-naive host- and you would be able to do this (get to most of them first) because you are sweeping rapidly through the whole population.
There isn't any evidence for 'blanket' protection after the first
infection. There is the concept of 'original antigenic sin' which has
been used to explain the disappearance of strains. After 10-20 years
of circulation of a strain, a new one emerges; upon infection the new
virus induces a memory immune response which is not effective against
the new strain, but which does neutralize older strains. Hence, the
older strains disappear. But it's probably too simplistic a view.
Fascinating- thanks for this. My understanding of the adaptive immune system is sketchy, but having looked up original antigenic sin (great name!) it is clear it could fit the bill as a plausible mechanism for strain replacement.
My other tack on the naive flu hosts angle was the possibility that a person might be particularly *extra-infectious* during their first ever flu infection- and that seasonal flus might be hanging on partially as a result of an extra large R0 ‘boost’ from such first ever infections- and that this could be snatched away from them by the rapid sweep of a new epidemic strain. Kids certainly seem to be very infectious (bit of an understatement!) but maybe that’s just kids generally, rather than kid’s first ever infections.
There isn't any evidence for 'blanket' protection after the first
infection. There is the concept of 'original antigenic sin' which has
been used to explain the disappearance of strains. After 10-20 years
of circulation of a strain, a new one emerges; upon infection the new
virus induces a memory immune response which is not effective against
the new strain, but which does neutralize older strains. Hence, the
older strains disappear. But it's probably too simplistic a view.
Fascinating- thanks for this. My understanding of the adaptive immune system is sketchy, but having looked up original antigenic sin (great name!) it is clear it could fit the bill as a plausible mechanism for strain replacement.
My other tack on the naive flu hosts angle was the possibility that a person might be particularly *extra-infectious* during their first ever flu infection- and that seasonal flus might be hanging on partially as a result of an extra large R0 ‘boost’ from such first ever infections- and that this could be snatched away from them by the rapid sweep of a new epidemic strain. Kids certainly seem to be very infectious (bit of an understatement!) but maybe that’s just kids generally, rather than kid’s first ever infections.